Juvéderm® XC

JUVÉDERM® XC in Peabody | Boston ,MA
What is JUVÉDERM® XC?
JUVÉDERM® XC is a dermal filler designed to fill moderate to severe facial wrinkles and folds, like your “smile lines” or “parentheses,” to create a smooth look. Smile lines, or parentheses, are the creases that run from the bottom of your nose to the corners of your mouth. The Juvederm lip is the most popular among all fillers.
JUVÉDERM® XC offers smooth and natural-looking results that last up to 1 year with optimal correction at initial treatment
Before the injection procedure, you will have a consultation with Dr. Shanthala. She will discuss whether JUVÉDERM® XC is appropriate for you, review your expectations and your desired results, as well as identify the areas to treat. She will also review any potential side effects.
JUVÉDERM® XC offers
- Provides Instant Results
- Is A Nonsurgical Treatment
- Provides Natural-Looking Results
- Lasts Up To 1 Year With Optimal Treatment
- Contains Modified Hyaluronic Acid (HA)
Before the injection procedure, you will have a consultation with Dr. Shanthala. She will discuss whether JUVÉDERM® XC is appropriate for you, review your expectations and your desired results, as well as identify the areas to treat. She will also review any potential side effects.
JUVÉDERM® XC is nonsurgical, and you will begin to see results instantly. You should be able to get back to your full routine after 24 hours
JUVÉDERM® XC contains lidocaine, a numbing agent designed to minimize pain that can occur during treatment. Dr. Shanthala may also choose to numb the treatment area with a topical numbing cream to further minimize pain.
Results are instant. JUVÉDERM® XC lasts up to 1 year with optimal correction at initial treatment.
Results are instant. JUVÉDERM® XC lasts up to 1 year with optimal correction at initial treatment.
Dr. Shanthala will discuss any potential side effects during the consultation. With JUVÉDERM® XC, the most common side effects are redness, swelling, tenderness, firmness, itching, lumps/bumps, discoloration, and bruising at the injection site.
JUVÉDERM® XC is nonsurgical, and you will begin to see results instantly. You should be able to get back to your full routine after 24 hours
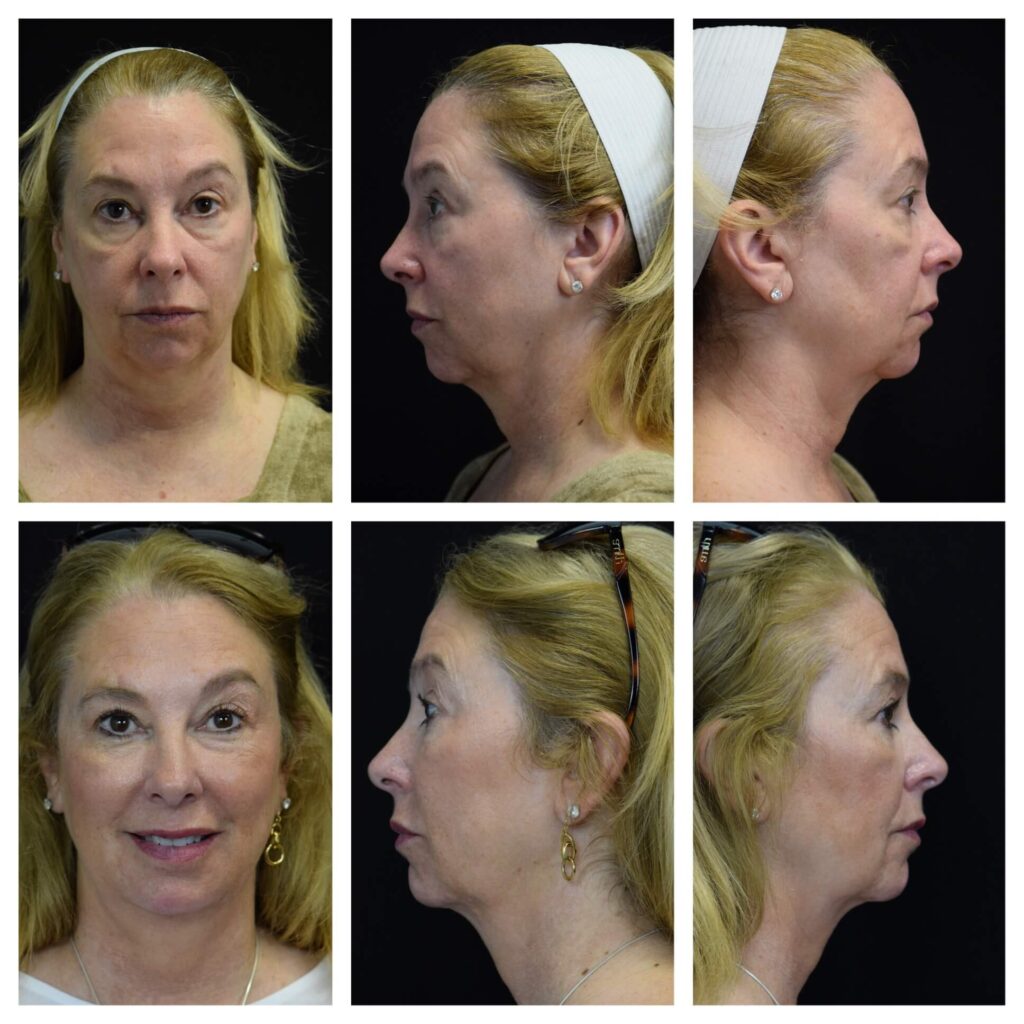
Before and after photos of JUVÉDERM® XC SHANTHALAMD

Reward Yourself Brilliantly
Brilliant Distinctions® allows you to earn points for savings on Allergan treatments such as BOTOX®, Juvéderm®, and Latisse®. If you’re looking for fillers such as JUVEDERM® XC performed by a physician in and around Peabody and Boston, request a consultation online, or contact us at (978) 854-5035 to schedule your appointment.

Dr. Shanthala Shivananjappa MD
Dr. Shanthala is a board-certified, Harvard trained anesthesiologist, assistant professor at Yale University, and staff at Massachusetts General Hospital prior to opening her cosmetic medicine clinic. Her extensive experience working with plastic surgical patients in the operating room for more than 25 years, paired with years of training in non-surgical cosmetic procedures, has enabled Dr. Shanthala to have a unique perspective on Anti-aging treatments.
